Title: 459P mRNA profile to predict platin sensitivity in NSCLC: A translational analysis of the ETOP/EORTC SPLENDOUR trial
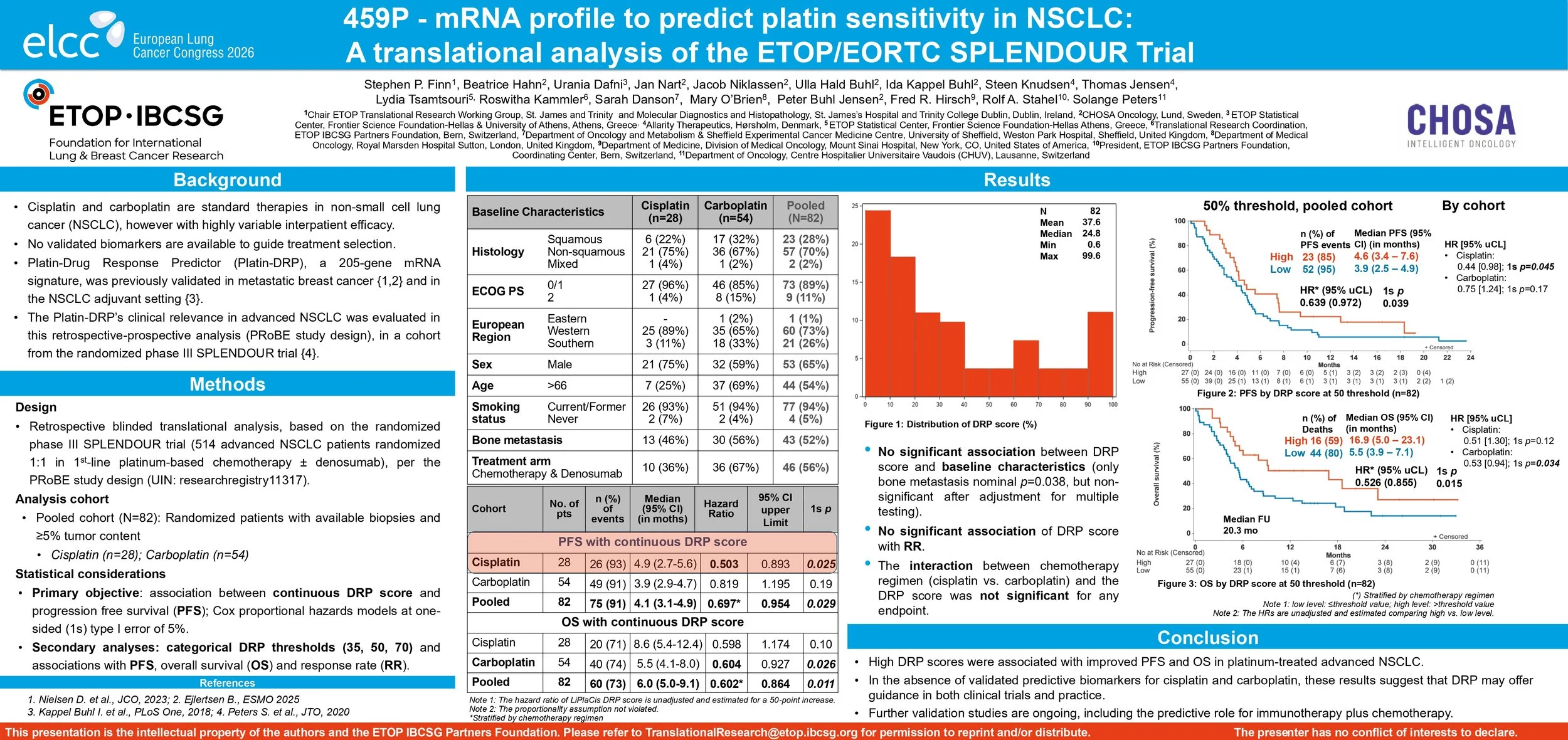
Background: Cisplatin and carboplatin are standard therapy in non-small cell lung cancer (NSCLC), but efficacy varies. Platin- Drug Response Predictor (Platin-DRP), a 205-gene mRNA signature, has shown clinical utility in two studies of metastatic breast cancer and two in adjuvant NSCLC. The Platin-DRP’s clinical relevance in advanced NSCLC was evaluated in this retrospective-prospective analysis (PRoBE study design), in a cohort from the randomized phase III SPLENDOUR trial (UIN: researchregistry11317).
Methods: SPLENDOUR randomized 514 advanced NSCLC patients 1:1 to platinum chemotherapy ± denosumab. The analysis cohort consisted of 82 patients with available biopsies and ≥5% tumor content (cisplatin n= 28, carboplatin n= 54). The primary objective was to assess the effect of the continuous DRP score on progression free survival (PFS) using Cox proportional hazards models at one-sided (1s) type I error of 5%. Secondary analyses evaluated categorical DRP thresholds (35, 50, 70) and associations with overall survival (OS) and response rate (RR).
Results: In the cohort of 82 patients, median for PFS was 4.1 months and for OS 6.0 months. In the cisplatin cohort, higher DRP scores were significantly associated with longer PFS (HR (per 50-point DRP increase)=0.50; 95% upper Confidence Limit (uCL): 0.89; 1s p = 0.025); In the carboplatin cohort, improved OS was significantly associated with higher DRP scores (HR (per 50-point increase)=0.60; 95% uCL: 0.93; 1s p=0.026). In the pooled cohort, higher continuous DRP scores correlated with improved PFS (HR = 0.70; 95% uCL: 0.95; 1s p=0.029) and longer OS (HR = 0.60; 95% uCL: 0.86; 1s p=0.011). Results were also significant in threshold-based analyses for PFS and OS (median OS 16.9 mo for score >50% vs 5.5 mo <50%, 1s p=0.015). No significant association was found between DRP and RR in any cohort.
Conclusions: High DRP scores were associated with improved PFS and OS in platinum-treated advanced NSCLC. In the absence of validated predictive biomarkers for cis- and carboplatin, these results suggest that DRP may offer guidance in both clinical trials and practice. Further validation studies are ongoing, including the predictive role for immunotherapy plus chemotherapy.
All authors and affiliations
Stephen P Finn (presenting author) (1), Beatrice Hahn: Hahn, B. (2) , Urania Dafni (3), Jan Nart: Nart, J. (2), Jacob Hansen Niklassen: Niklassen, J.H. (2) , Ulla Hald Buhl: Buhl, U.H. (2), Ida Kappel Buhl: Buhl, I.K. (2) , Steen Knudsen: Knudsen, S. (4) , Thomas Jensen: Jensen, T. (4) , Lydia Tsamtsouri (5), Roswitha Kammler (6) , Sarah Danson (7), Mary O’Brien (8), Peter Buhl Jensen: Jensen, P.B. (2), Fred R. Hirsch (9), Rolf A. Stahel (10), Solange Peters (11)
(1) Chair ETOP Translational Research Working Group, St. James and Trinity and Molecular Diagnostics and Histopathology, St. James’s Hospital and Trinity College Dublin, Dublin, Ireland
(2) CHOSA Oncology, Lund, Sweden
(3) ETOP Statistical Center, Frontier Science Foundation-Hellas & University of Athens, Athens, Greece ,
(4) Allarity Therapeutics, Hørsholm, Denmark
(5) ETOP Statistical Center, Frontier Science Foundation-Hellas Athens, Greece,
(6) Translational Research Coordination, ETOP IBCSG Partners Foundation, Bern, Switzerland
(7) Department of Oncology and Metabolism & Sheffield Experimental Cancer Medicine Centre, University of Sheffield, Weston Park Hospital, Sheffield, United Kingdom
(8) Department of Medical Oncology, Royal Marsden Hospital Sutton, London, United Kingdom
(9) Department of Medicine, Division of Medical Oncology, Mount Sinai Hospital, New York, CO, United States of America
(10) President, ETOP IBCSG Partners Foundation, Coordinating Center, Bern, Switzerland
(11) Department of Oncology, Centre Hospitalier Universitaire Vaudois (CHUV), Lausanne, Switzerland
Contact information:
Peter Buhl Jensen (CHOSA Oncology, CEO): peter@chosa.bio